Surface Finishes
Electropolishing Services
Electropolishing is an electrochemical process utilized to enhance the surface area coating of a part by removing material to level microscopic optimal and valleys.
ECOREPRAP Electropolishing Service
At ECOREPRAP, we offer electropolishing as a precision surface finishing process for stainless steel and other compatible alloys. This electrochemical process removes a controlled amount of material from the surface, reducing micro-peaks and burrs to achieve a smoother, cleaner, and more uniform finish.
Electropolishing is particularly effective in improving surface roughness, enhancing corrosion resistance, and reducing the risk of contamination. By eliminating embedded particles and surface irregularities, it creates a passive, chromium-rich surface layer that is well-suited for applications in medical, food processing, and high-purity environments.
Unlike mechanical polishing, electropolishing can reach complex geometries, internal passages, and hard-to-access features without introducing mechanical stress or deformation. This makes it ideal for precision CNC machined parts with intricate designs.
Our process includes controlled cleaning, electrochemical treatment, rinsing, and passivation to ensure consistent and repeatable results. We work closely with part geometry, initial surface condition, and functional requirements to achieve the desired surface quality and performance.
With our machining background, we understand how electropolishing interacts with upstream processes, allowing us to better control final surface finish, dimensional impact, and overall part functionality.
| Applicable Materials | Cosmetic Availability | Visual Appearance |
| Stainless steel, Aluminum, Copper, Brass | NA | Smooth, glossy finish |
Electropolishing Parts


Electropolishing Process
Here’s how to apply brushing and electropolishing:
- The Part’s surface is cleaned and prepared by power brushing.
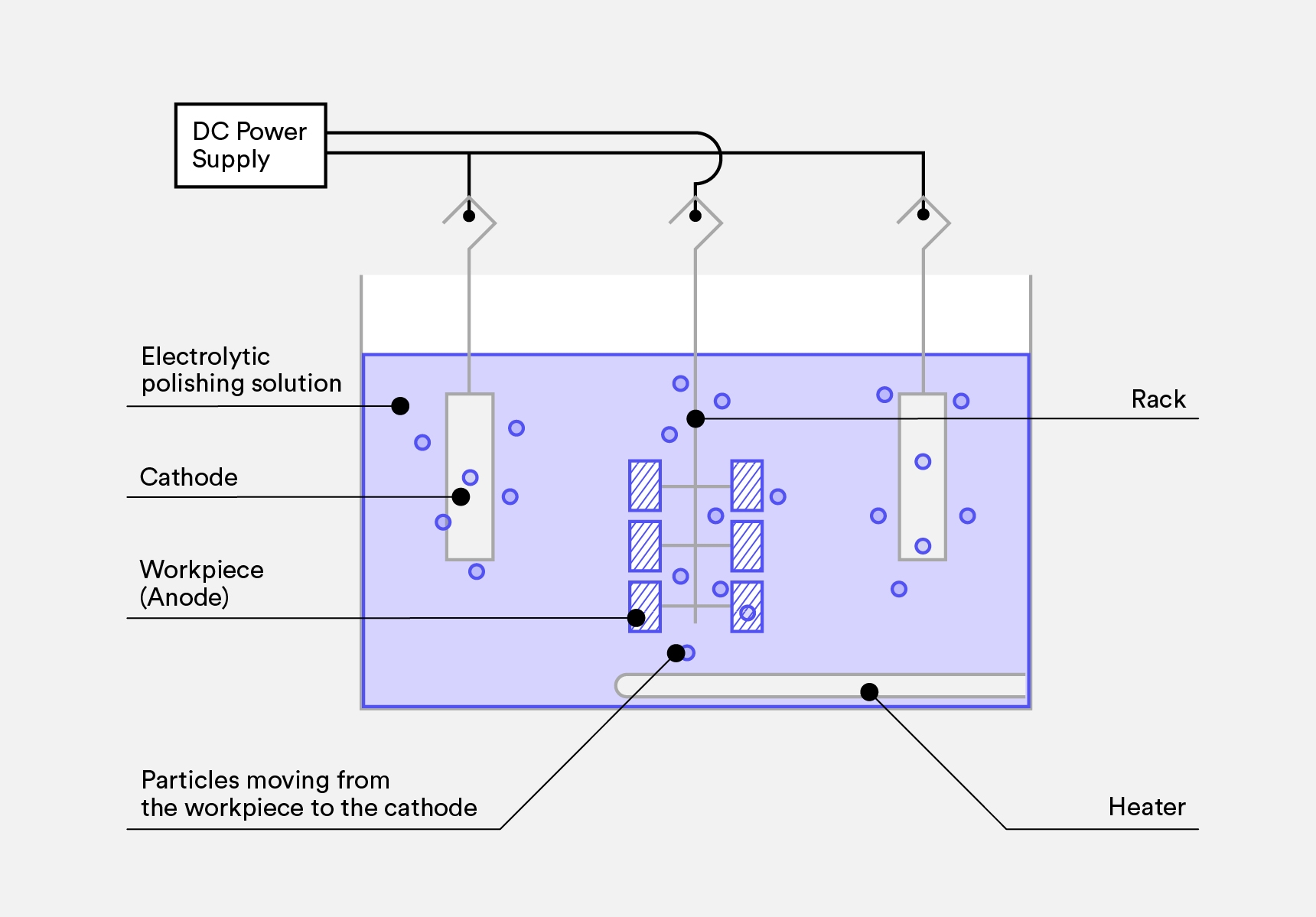
- The metal part being electropolished (aka anode) is connected to the positive terminal of a direct current (DC) rectifier and submerged in a tank of an electrolyte solution. The unfavorable terminal of the rectifier is connected to a cathode (typically stainless steel) that is likewise submerged in the tank.
- A current runs through the solution from the anode to the cathode, causing ions on the surface of the material to oxidize and dissolve into the solution.
- Parts are then rinsed from the electrolyte solution in a different tank.
- To finalize the finish, parts go through a final post-dip in a Nitric dip or a Citric acid, rinsed with cold water and are put to dry.
Electropolishing Design Considerations
FAQs
Electropolishing is an electrochemical process that removes a controlled amount of surface material to improve the finish, corrosion resistance, and cleanliness of metal parts.
Electropolishing enhances the appearance, passivates the surface, and improves corrosion resistance, making it an attractive finish for stainless steel and other metals.
Electropolishing is primarily used for stainless steel, but it can also be applied to other metals such as aluminum, copper, and brass.
Electropolishing typically removes a very thin layer of material, resulting in minimal dimensional changes, suitable for precision components.
Yes, electropolishing is effective on parts with complex shapes, fine details, and intricate geometries.
Electropolishing can be combined with other processes, such as passivation or coating, to achieve specific performance and appearance requirements.
Electropolishing improves the electrical conductivity of metal parts and does not provide electrical insulation.
Post-electropolishing treatments like passivation may be required for specific applications to enhance corrosion resistance.
Electropolishing can be environmentally friendly when using eco-friendly electrolytes and efficient wastewater treatment.
Electropolishing produces a smooth, bright, and reflective surface finish with reduced micro-roughness, typically with an Ra value of less than 0.2 micrometers (渭m).